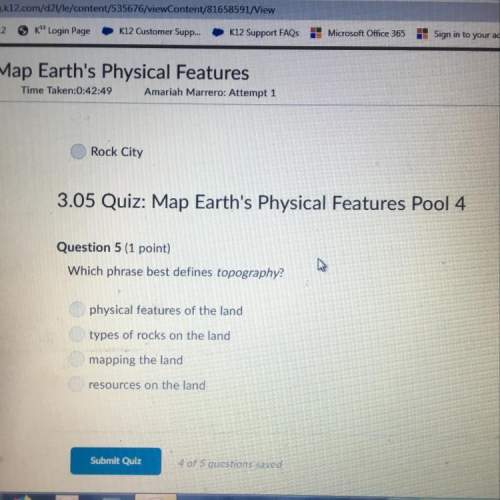
Chemistry, 23.09.2020 14:01 fespinoza019
When of a certain molecular compound X are dissolved in of benzonitrile , the freezing point of the solution is measured to be . Calculate the molar mass of X. If you need any additional information on benzonitrile, use only what you find in the ALEKS Data resource. Also, be sure your answer has a unit symbol, and is rounded to significant digit.

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 22:00
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3

Chemistry, 22.06.2019 01:30
Agas is contained in a thick walled balloon when the pressure changes from 1.21 atm to 2.52 the volume changes from 3.75 l to 1.72 l and the temperature change from 293k to blank k
Answers: 3

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1
You know the right answer?
When of a certain molecular compound X are dissolved in of benzonitrile , the freezing point of the...
Questions

Mathematics, 19.05.2020 18:03

History, 19.05.2020 18:03




English, 19.05.2020 18:03




Mathematics, 19.05.2020 18:03


Social Studies, 19.05.2020 18:03





Chemistry, 19.05.2020 18:03


Mathematics, 19.05.2020 18:03