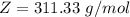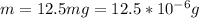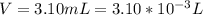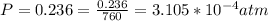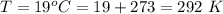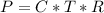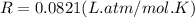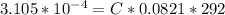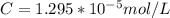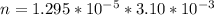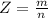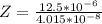Working on-board a research vessel somewhere at sea, you have (carefully) isolated 12.5 micrograms (12.5 ×10–6 g) of what you hope is pure saxitoxin (a non-electrolyte) from a poisonous (and quite cross) puffer fish. You dissolve this sample in 3.10 mL of water and determine that the osmotic pressure of the resulting solution is 0.236 torr at 19ºC (760 torr = 1.00 atm). What is the molar mass of the compound?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which of the following is a compound? a.carbon b.oxygen c.hydrogen d.water
Answers: 2

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1
You know the right answer?
Working on-board a research vessel somewhere at sea, you have (carefully) isolated 12.5 micrograms (...
Questions


History, 29.10.2019 15:31

Law, 29.10.2019 15:31


Chemistry, 29.10.2019 15:31

History, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31


Mathematics, 29.10.2019 15:31


History, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31

Social Studies, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31




Mathematics, 29.10.2019 15:31

Mathematics, 29.10.2019 15:31