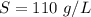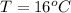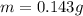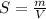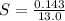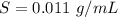Chemistry, 22.09.2020 21:01 ncontreras06
A geochemist in the field takes a 13.0 mL sample of water from a rock pool lined with crystals of a certain mineral compound X. He notes the temperature of the pool, 16.° C, and caps the sample carefully. Back in the lab, the geochemist filters the sample and then evaporates all the water under vacuum. Crystals of X are left behind. The researcher washes, dries and weighs the crystals. They weigh 0.143 g
Required:
Using only the information above, can you calculate the solubility of X in water at 15°C ? If you said yes, calculate it.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1


Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 22:30
Why is it possible for different microorganisms to extract energy not only from carbohydrates and other biological molecules but from a large variety of substances?
Answers: 1
You know the right answer?
A geochemist in the field takes a 13.0 mL sample of water from a rock pool lined with crystals of a...
Questions

Biology, 22.12.2019 03:31


English, 22.12.2019 03:31







Health, 22.12.2019 03:31



Biology, 22.12.2019 03:31


Advanced Placement (AP), 22.12.2019 03:31


Mathematics, 22.12.2019 03:31

English, 22.12.2019 03:31