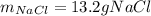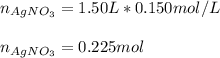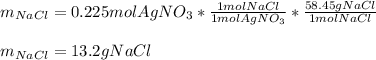Chemistry, 22.09.2020 01:01 emma042902
Calculate the mass of NaCl that must be added to completely react with 1.50 L of a 0.150 M AgNO3 solution according to the reaction below: NaCl (aq) + AgNO3 (aq) → AgCl (s) + NaNO3 (aq)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1


Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1
You know the right answer?
Calculate the mass of NaCl that must be added to completely react with 1.50 L of a 0.150 M AgNO3 sol...
Questions


History, 20.08.2019 04:30

Physics, 20.08.2019 04:30

History, 20.08.2019 04:30

Mathematics, 20.08.2019 04:30

Biology, 20.08.2019 04:30



Social Studies, 20.08.2019 04:30


Arts, 20.08.2019 04:30

History, 20.08.2019 04:30


History, 20.08.2019 04:30

Mathematics, 20.08.2019 04:30



Mathematics, 20.08.2019 04:30


Social Studies, 20.08.2019 04:30