
Chemistry, 20.09.2020 15:01 smithsavannah295
olecular iodine, I2(g), dissociates into iodine atoms at 625 K with a first-order rate constant of 0.271 s-1. (a) What is the half-life for this reaction? s (b) If you start with 0.048 M I2 at this temperature, how much will remain after 5.37 s assuming that the iodine atoms do not recombine to form I2? M

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 15:00
Why does a plastic bottle that is sealed at a high altitude change it’s shape when taken to lower altitude
Answers: 2


Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
olecular iodine, I2(g), dissociates into iodine atoms at 625 K with a first-order rate constant of 0...
Questions



Mathematics, 27.06.2019 13:00


Biology, 27.06.2019 13:00

Arts, 27.06.2019 13:00

Mathematics, 27.06.2019 13:00







English, 27.06.2019 13:00

English, 27.06.2019 13:00


Chemistry, 27.06.2019 13:00


English, 27.06.2019 13:00

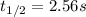
![[I_2]=0.011M](/tpl/images/0771/9802/19391.png)
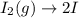
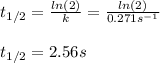
![[I_2]=[I_2]_0exp(-kt)](/tpl/images/0771/9802/e9e96.png)
![[I_2]=0.048Mexp(-0.271s^{-1}*5.37s)\\\\](/tpl/images/0771/9802/4a6e2.png)



