A common issue in organic chemistry processes is trace contamination. Organic solvents can become contaminated with water. One method of removing the water contamination is to freeze dry the solvent. Unfortunately, not all solvents, like acetonitrile, are compatible with this method. Another method is to add a chemical to react with the water. One such chemical is calcium hydride, which will react to form calcium hydroxide and hydrogen gas. When you checked the chemical inventory, you realized you only had 24.6g of calcium hydride on hand. If you had a 1.5L container of organic solvent that contained 14.0g of water, what will be the outcome of your attempt to dewater the solvent?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3
You know the right answer?
A common issue in organic chemistry processes is trace contamination. Organic solvents can become co...
Questions



Advanced Placement (AP), 27.07.2019 12:30

World Languages, 27.07.2019 12:30


Biology, 27.07.2019 12:30

English, 27.07.2019 12:30



English, 27.07.2019 12:30

Biology, 27.07.2019 12:30


History, 27.07.2019 12:30


History, 27.07.2019 12:30

Mathematics, 27.07.2019 12:30

Mathematics, 27.07.2019 12:30



Geography, 27.07.2019 12:30

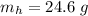
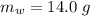
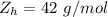
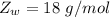
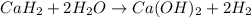
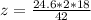
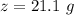 of water
of water