
Chemistry, 20.09.2020 07:01 EricaLSH7624
The element X has three naturally occurring isotopes. The isotopic masses (amu) and % abundances of the isotopes are given in the table below. Calculate the average atomic mass of the element in amu. PLEASE HELP ASAP THANK YOU SO MUCH
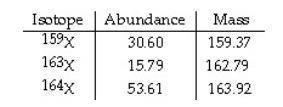

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Supongamos que estás estudiando dos estrellas. ambas estrellas tienen la misma magnitud aparente, pero la estrella a tiene una magnitud absoluta mayor que la estrella b. ¿que puedes decir acerca de la distancia a la tierra de estas dos estrellas?
Answers: 3

Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1
You know the right answer?
The element X has three naturally occurring isotopes. The isotopic masses (amu) and % abundances of...
Questions

Social Studies, 19.07.2019 07:50

Biology, 19.07.2019 07:50

Social Studies, 19.07.2019 07:50

History, 19.07.2019 07:50

History, 19.07.2019 07:50

Business, 19.07.2019 07:50

Biology, 19.07.2019 07:50

English, 19.07.2019 07:50

English, 19.07.2019 07:50

English, 19.07.2019 07:50

English, 19.07.2019 07:50



Mathematics, 19.07.2019 07:50



Geography, 19.07.2019 07:50

English, 19.07.2019 07:50


Biology, 19.07.2019 07:50



