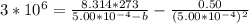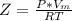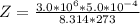Chemistry, 10.09.2020 04:01 llnapier8924
A certain gas obeys the van der Waals equation with a = 0.50 m6 Pa mol−2. Its molar volume is found to be 5.00 × 10–4 m3 mol−1 at 273 K and 3.0 MPa. From this information calculate the van der Waals constant b. What is the compression factor for this gas at the prevailing temperature and pressure?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 21:30
Under which circumstances are kp and kc equal for the reaction aa(g)+bb(g)⇌cc(g)+dd(g)?
Answers: 2

Chemistry, 23.06.2019 02:50
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3

Chemistry, 23.06.2019 03:30
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1
You know the right answer?
A certain gas obeys the van der Waals equation with a = 0.50 m6 Pa mol−2. Its molar volume is found...
Questions






World Languages, 19.10.2019 07:50



Mathematics, 19.10.2019 07:50

Mathematics, 19.10.2019 07:50

Social Studies, 19.10.2019 07:50


Mathematics, 19.10.2019 07:50


Advanced Placement (AP), 19.10.2019 07:50





Biology, 19.10.2019 07:50

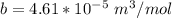
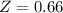
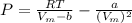
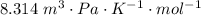 for R , 273K for T ,
for R , 273K for T , 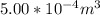 for
for 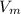 ,
, 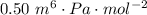 for a and
for a and 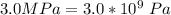 for P
for P