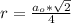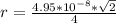The density of lead, which has the FCC structure, is 11.36 . The atomic weight of lead is 207.19 . Use Avogadro's number: 6.02210. Calculatethe lattice parameter(Enter your answer to three significant figures.) = 2.75*10^21 the atomic radius of lead(Enter your answer to three significant figures.) =

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 22:30
What if it is did darwin used to support his theory of evolution
Answers: 1

Chemistry, 23.06.2019 00:00
How do you determine the percent yield of a chemical reaction
Answers: 1

Chemistry, 23.06.2019 02:00
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2
You know the right answer?
The density of lead, which has the FCC structure, is 11.36 . The atomic weight of lead is 207.19 . U...
Questions



Biology, 04.09.2019 19:30







History, 04.09.2019 19:30










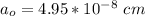
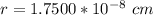
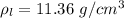
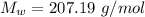
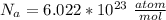
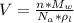
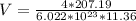
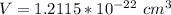
![a_o = \sqrt[3]{V }](/tpl/images/0748/9324/a210e.png)
![a_o = \sqrt[3]{1.211 5 *10^{-22} }](/tpl/images/0748/9324/32761.png)