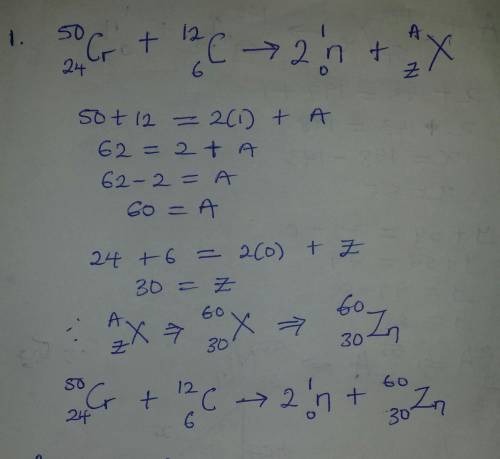Chemistry, 08.09.2020 14:01 nerdypineapple
Select the product of the following nuclear reaction: 5024Cr+12 6C→2 10n + ? Express your answer as an element symbol including subscripts and superscripts.
if you had a high-energy xenon particle beam and wanted to make gold, what element would you bombard?
? +143 54Xe→197 79Au+10n
Express your answer as an element symbol including subscripts and superscripts.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

You know the right answer?
Select the product of the following nuclear reaction: 5024Cr+12 6C→2 10n + ? Express your answer as...
Questions


English, 19.09.2019 13:50



Geography, 19.09.2019 13:50

Mathematics, 19.09.2019 13:50



Health, 19.09.2019 13:50

Spanish, 19.09.2019 13:50

Chemistry, 19.09.2019 13:50


Biology, 19.09.2019 13:50


Biology, 19.09.2019 13:50

English, 19.09.2019 13:50

History, 19.09.2019 13:50


English, 19.09.2019 13:50

Biology, 19.09.2019 13:50