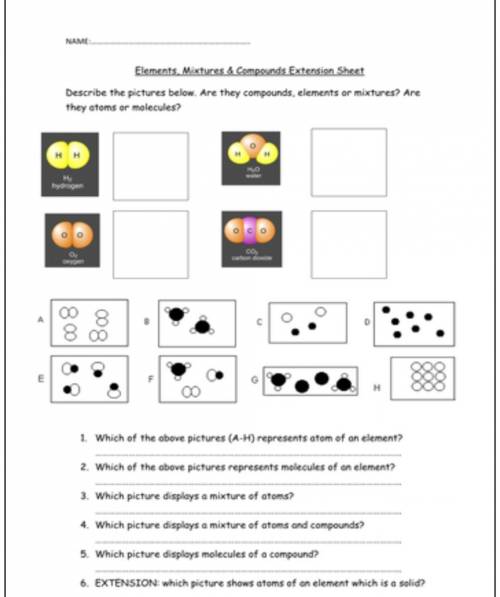
Hello
Please solve this worksheet
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

You know the right answer?
Questions




Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10



Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10

History, 27.04.2021 19:10

French, 27.04.2021 19:10







Mathematics, 27.04.2021 19:10