
Chemistry, 31.08.2020 23:01 eskarletche8
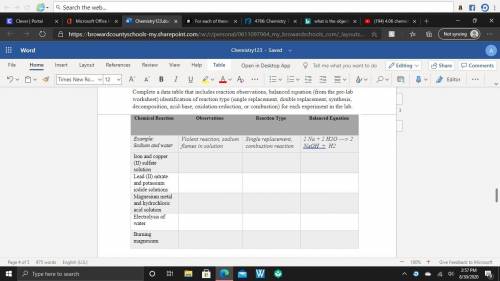
Data: Complete a data table that includes reaction observations, balanced equation (from the pre-lab worksheet) identification of reaction type (single replacement, double replacement, synthesis, decomposition, acid-base, oxidation-reduction, or combustion) for each experiment in the lab. Lead (II) nitrate and potassium iodide solutions Magnesium metal and hydrochloric acid solution Electrolysis of water Burning magnesium

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
You know the right answer?
Data: Complete a data table that includes reaction observations, balanced equation (from the pre-lab...
Questions

English, 14.11.2020 01:00






Mathematics, 14.11.2020 01:00



Arts, 14.11.2020 01:00

SAT, 14.11.2020 01:00


Mathematics, 14.11.2020 01:00

Mathematics, 14.11.2020 01:00


Computers and Technology, 14.11.2020 01:00


English, 14.11.2020 01:00




