
Chemistry, 31.08.2020 02:01 sarrivera579
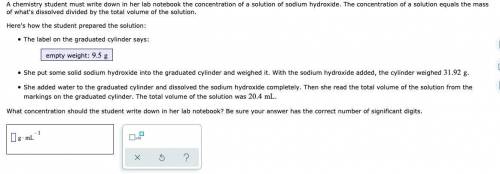
A chemistry student must write down in her lab notebook the concentration of a solution of sodium hydroxide. The concentration of a solution equals the mass of what's dissolved divided by the total volume of the solution. Here's how the student prepared the solution: The label on the graduated cylinder says: empty weight: She put some solid sodium hydroxide into the graduated cylinder and weighed it. With the sodium hydroxide added, the cylinder weighed . She added water to the graduated cylinder and dissolved the sodium hydroxide completely. Then she read the total volume of the solution from the markings on the graduated cylinder. The total volume of the solution was . What concentration should the student write down in her lab notebook? Be sure your answer has the correct number of significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

You know the right answer?
A chemistry student must write down in her lab notebook the concentration of a solution of sodium hy...
Questions


Chemistry, 07.10.2020 14:01



History, 07.10.2020 14:01

Mathematics, 07.10.2020 14:01

Chemistry, 07.10.2020 14:01

Mathematics, 07.10.2020 14:01


Mathematics, 07.10.2020 14:01

History, 07.10.2020 14:01





Mathematics, 07.10.2020 14:01


Mathematics, 07.10.2020 14:01




