
Chemistry, 30.08.2020 14:01 jesussanchez1445
What is the pH of a buffer made from 0.130 mol of HCNO (Ka = 3.5 × 10⁻⁴) and 0.410 mol of NaCNO in 2.0 L of solution?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1
You know the right answer?
What is the pH of a buffer made from 0.130 mol of HCNO (Ka = 3.5 × 10⁻⁴) and 0.410 mol of NaCNO in 2...
Questions

Mathematics, 18.03.2021 01:20

Mathematics, 18.03.2021 01:20


Mathematics, 18.03.2021 01:20

History, 18.03.2021 01:20

History, 18.03.2021 01:20

English, 18.03.2021 01:20


Mathematics, 18.03.2021 01:20



Mathematics, 18.03.2021 01:20


Chemistry, 18.03.2021 01:20

History, 18.03.2021 01:20

Mathematics, 18.03.2021 01:20

Advanced Placement (AP), 18.03.2021 01:20



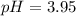
![pH=pKa+log(\frac{[base]}{[acid]} )](/tpl/images/0734/8563/33848.png)
![[base]=\frac{0.410mol}{2.0L}=0.205M](/tpl/images/0734/8563/eabfc.png)
![[acid]=\frac{0.130mol}{2.0L}=0.065M](/tpl/images/0734/8563/e375b.png)



