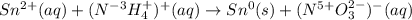Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which of the following pairs of elements belong to the same groupa. h and he b. li and bec. c and pb d. ga and ge
Answers: 1

Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2


Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1
You know the right answer?
What element is being oxidized in the following redox reaction? Sn 2+(aq) + NH4 +(aq) → Sn(s) + NO3...
Questions


Social Studies, 09.07.2019 13:50








Social Studies, 09.07.2019 13:50


Chemistry, 09.07.2019 13:50


History, 09.07.2019 13:50

Biology, 09.07.2019 13:50


Social Studies, 09.07.2019 13:50

Mathematics, 09.07.2019 13:50

Chemistry, 09.07.2019 13:50