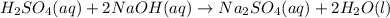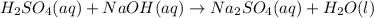Chemistry, 29.08.2020 15:01 johnsonkia873
Sulfuric acid, H2SO4, can be neutralized by sodium hydroxide, NaOH. The unbalanced equation is: H2SO4(aq) + NaOH(aq) → Na2SO4(aq) + H2O(l) A student who was asked to balance the reaction wrote the following: H2SO4(aq) + Na2OH(aq) →Na2SO4(aq) + H3O(l) Is this correct? Explain why or why not using what you know about the law of conservation of mass and chemical changes. If necessary, provide the correct balanced equation.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Zinc + lead(ii) nitrate yield zinc nitrate + leadwhat's the chemical equation for this?
Answers: 1

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
Sulfuric acid, H2SO4, can be neutralized by sodium hydroxide, NaOH. The unbalanced equation is: H2SO...
Questions

History, 30.04.2021 14:00

Social Studies, 30.04.2021 14:00

Chemistry, 30.04.2021 14:00


Mathematics, 30.04.2021 14:00

Computers and Technology, 30.04.2021 14:00

Computers and Technology, 30.04.2021 14:00

Health, 30.04.2021 14:00

Biology, 30.04.2021 14:00


Business, 30.04.2021 14:00




Physics, 30.04.2021 14:00

English, 30.04.2021 14:00

Mathematics, 30.04.2021 14:00


Geography, 30.04.2021 14:00

Mathematics, 30.04.2021 14:00