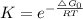Chemistry, 28.08.2020 16:01 j1theking18
Choose the best answer below. Which of the following reactions will have the largest equilibrium constant at 298 K?
a) 302(g) → 203(9) AGOrxn = +326 kJ
b) Mg(s) + N20(g) → Mgo(s) + N2(g) AG9rxn = -673.0 kJ
c) 2Hg(g) + O2(g) → 2HgO(s) AGºrx = -180.8 kJ
d) CaCO3(s) » Cao(s) + CO2(g) AG = +131.1 kJ
It is not possible to determine the reaction with the largest equilibrium constant using the given information.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 00:10
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2

You know the right answer?
Choose the best answer below. Which of the following reactions will have the largest equilibrium con...
Questions



Business, 14.12.2020 23:40


English, 14.12.2020 23:40


Mathematics, 14.12.2020 23:40



Mathematics, 14.12.2020 23:40


Physics, 14.12.2020 23:40

English, 14.12.2020 23:40

Mathematics, 14.12.2020 23:40


Mathematics, 14.12.2020 23:40


Chemistry, 14.12.2020 23:40


Mathematics, 14.12.2020 23:40