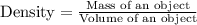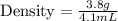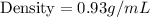Chemistry, 28.08.2020 02:01 sarahhfaithhh
Calculate the density of an object with a mass of 3.8 g, that when placed in a 10.0 mL graduated cylinder with an initial volume of 4.5 mL rises to a final volume of 8.6 mL. *

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1
You know the right answer?
Calculate the density of an object with a mass of 3.8 g, that when placed in a 10.0 mL graduated cyl...
Questions


Social Studies, 04.05.2021 01:00


Mathematics, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00


Social Studies, 04.05.2021 01:00






History, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00

Social Studies, 04.05.2021 01:00

Mathematics, 04.05.2021 01:00