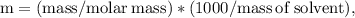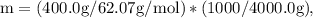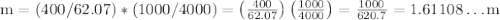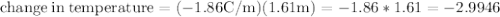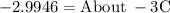Chemistry, 26.08.2020 23:01 keigleyhannah30
Ethylene glycol (C2H6O2) is used as an antifreeze in cars. If 400 g of ethylene glycol is added to 4.00 kg of water, what is the molality? Calculate how much the freezing point of water will be lowered. The freezing-point depression constant for water is Kf = –1.86°C/m.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 23.06.2019 02:30
Which of the four hypothetical substances you investigated would be most harmful to living organisms? 50 points!
Answers: 2

You know the right answer?
Ethylene glycol (C2H6O2) is used as an antifreeze in cars. If 400 g of ethylene glycol is added to 4...
Questions

SAT, 18.11.2020 03:50

Mathematics, 18.11.2020 03:50


Arts, 18.11.2020 03:50

SAT, 18.11.2020 03:50

Mathematics, 18.11.2020 03:50


Mathematics, 18.11.2020 03:50




Mathematics, 18.11.2020 03:50


English, 18.11.2020 03:50

History, 18.11.2020 03:50

Chemistry, 18.11.2020 03:50

Mathematics, 18.11.2020 03:50

Mathematics, 18.11.2020 03:50

History, 18.11.2020 03:50

English, 18.11.2020 03:50