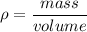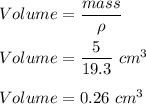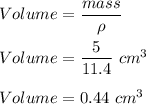Chemistry, 27.08.2020 20:01 mckinley2006
The density of gold is 19.3 g/cm3 and the density of lead is 11.4 g/cm3. If you have 5 grams of gold and 5 grams of lead, which will have the greater volume?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1

Chemistry, 23.06.2019 05:30
Astudent made the lewis dot diagram of a compound as shown. mg is written with two dots shown on its top. an o is written on each side of mg. each o has six dots around it. an arrow is shown from one dot on mg toward the vacant space around the o on the right. another arrow is shown from the other dot on mg toward the vacant space around the o on the left. the title of the art is students lewis dot model. what is the error in the lewis dot diagram? an o atom should transfer all its six electrons to mg because the formula is mgo. both electrons of mg should be transferred to one o atom because the formula is mgo. the electrons should be transferred from each o atom to mg because mg has fewer electrons. the number of dots around mg should be four because it has to transfer two electrons to each o.
Answers: 2

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
The density of gold is 19.3 g/cm3 and the density of lead is 11.4 g/cm3. If you have 5 grams of gold...
Questions

Mathematics, 16.01.2020 17:31


Mathematics, 16.01.2020 17:31

Mathematics, 16.01.2020 17:31


Physics, 16.01.2020 17:31

History, 16.01.2020 17:31






Computers and Technology, 16.01.2020 17:31

Social Studies, 16.01.2020 17:31

Social Studies, 16.01.2020 17:31



Computers and Technology, 16.01.2020 17:31


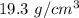 and the density of lead is
and the density of lead is 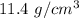 respectively .
respectively .