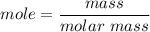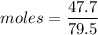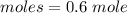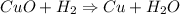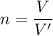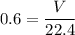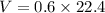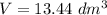Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
If a 12-v battery is connected to a circuit that has a current of 3.0 a, what is the total resistance in the circuit? 36 ohms 4 ohms 0.25 ohms
Answers: 1

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
How many dm³ of hydrogen, measured at s. t.p.,would be needed to reduce 47.7g of copper(II) oxide to...
Questions


Mathematics, 12.11.2021 14:00

History, 12.11.2021 14:00



Biology, 12.11.2021 14:00

English, 12.11.2021 14:00


History, 12.11.2021 14:00


Mathematics, 12.11.2021 14:00


History, 12.11.2021 14:00

Mathematics, 12.11.2021 14:00

Mathematics, 12.11.2021 14:00


English, 12.11.2021 14:00


Mathematics, 12.11.2021 14:00