Chemistry, 24.08.2020 22:01 templetiggerxyz
You have 50 milliliters of a 0.45 M MgBr2 solution and dilute it to a total volume of 300 milliliters. What is the molarity of the resulting solution? Describe in words how you will find the new molarity.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2


Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
You know the right answer?
You have 50 milliliters of a 0.45 M MgBr2 solution and dilute it to a total volume of 300 milliliter...
Questions



Social Studies, 26.07.2019 08:30



Mathematics, 26.07.2019 08:30

Physics, 26.07.2019 08:30



History, 26.07.2019 08:30

English, 26.07.2019 08:30

Social Studies, 26.07.2019 08:30

Geography, 26.07.2019 08:30


Geography, 26.07.2019 08:30




Mathematics, 26.07.2019 08:30

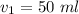 .
.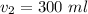 .
.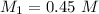 .
.