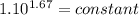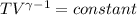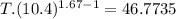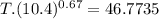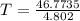Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3
You know the right answer?
An ideal gas with γ = 1.67 has an initial temperature of 0°C, initial volume of 10.0 liters, and ini...
Questions


History, 04.01.2022 04:20

SAT, 04.01.2022 04:20






English, 04.01.2022 04:20

Mathematics, 04.01.2022 04:20



Biology, 04.01.2022 04:20

English, 04.01.2022 04:20


Arts, 04.01.2022 04:20

Mathematics, 04.01.2022 04:20



Social Studies, 04.01.2022 04:20

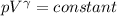 .
.