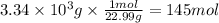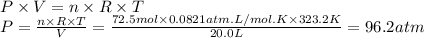Solid sodium reacts with liquid water to form hydrogen gas according to the equation 2 Na(s) + 2 H₂O(l) → 2 NaOH(aq) + H₂(g) What is the pressure of hydrogen gas in the 20.0 L headspace of a reactor vessel when 3.34 kg sodium is reacted with excess water at 50.0°C?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 06:30
Type the correct answer in the box. spell all words correctly.what is the correct term for living the most sustainable life you can within your current circumstances? when your are being as sustainable as you can within your current lifestyle, you are said to be sustainability.
Answers: 3

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1
You know the right answer?
Solid sodium reacts with liquid water to form hydrogen gas according to the equation 2 Na(s) + 2 H₂O...
Questions

Mathematics, 05.02.2021 21:20


Geography, 05.02.2021 21:20


History, 05.02.2021 21:20

Mathematics, 05.02.2021 21:20

Mathematics, 05.02.2021 21:20


Physics, 05.02.2021 21:20



Mathematics, 05.02.2021 21:20





English, 05.02.2021 21:20


Biology, 05.02.2021 21:20