
Chemistry, 19.08.2020 02:01 markuswalter1043
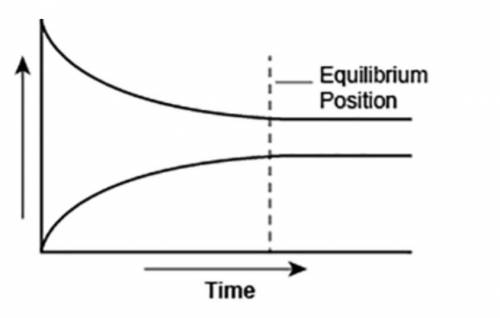
A student made a graph to show the chemical equilibrium position of a reaction.
The student forgot to label the y-axis of the graph.
What best explains the label that the student should use on the y-axis? A. Concentration, because as the amount of product decreases, the amount of reactant increases over time.
B. Reaction rate, as the rates of forward and backward reactions become equal at equilibrium.
C. Concentration, because the amounts of reactants and products remain constant after equilibrium is reached.
D. Reaction rate, as the rate of forward reaction increases and rate of backward reaction decreases over time.

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
A student made a graph to show the chemical equilibrium position of a reaction.
The student forgot...
Questions

Geography, 29.09.2019 09:30

Mathematics, 29.09.2019 09:30




Mathematics, 29.09.2019 09:30

Social Studies, 29.09.2019 09:30

Mathematics, 29.09.2019 09:30

Mathematics, 29.09.2019 09:30

Physics, 29.09.2019 09:50

Social Studies, 29.09.2019 09:50

Social Studies, 29.09.2019 09:50

Physics, 29.09.2019 09:50




History, 29.09.2019 09:50

History, 29.09.2019 09:50

History, 29.09.2019 09:50



