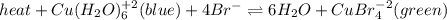The following equilibrium is formed when copper and bromide ions are placed in a solution:
heat + Cu(H2O)6 ^+2 (blue) + 4Br- <--> 6H2O + CuBr4^-2 (green)
A) answer the following questions when KBr is added to the solution:
1. What will happen to the equilibrium?
2. What will be the color of the solution?
3. Will the solution be hotter or cooler? Explain.
B) What will be the color of the solution when the solution is heated?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
The following equilibrium is formed when copper and bromide ions are placed in a solution:
heat + C...
Questions


Mathematics, 09.03.2021 04:10





Advanced Placement (AP), 09.03.2021 04:10

History, 09.03.2021 04:10

English, 09.03.2021 04:10


Mathematics, 09.03.2021 04:10

Mathematics, 09.03.2021 04:10

Mathematics, 09.03.2021 04:10



Mathematics, 09.03.2021 04:10


Business, 09.03.2021 04:10