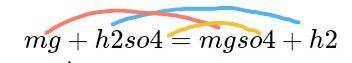Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
Balance equation for Magnesium + Sulphuric acid = Magnesium sulphate + Hydrogen gas...
Questions

Mathematics, 13.11.2020 19:20

Advanced Placement (AP), 13.11.2020 19:20

Mathematics, 13.11.2020 19:20


Mathematics, 13.11.2020 19:20


Biology, 13.11.2020 19:20









Biology, 13.11.2020 19:20




English, 13.11.2020 19:20

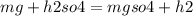 ( hydrogen gas)
( hydrogen gas)