
Chemistry, 16.08.2020 14:01 animaljamissofab
In the following net ionic equation, identify each reactant as either a Bronsted-Lowry acid or a Bronsted-Lowry base. HCN(aq) H2O(l) CN-(aq) H3O (aq) B-L B-L
The formula of the reactant that acts as a proton donor is
The formula of the reactant that acts as a proton acceptor is

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 04:30
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1
You know the right answer?
In the following net ionic equation, identify each reactant as either a Bronsted-Lowry acid or a Bro...
Questions



History, 24.08.2019 05:00

History, 24.08.2019 05:00



Mathematics, 24.08.2019 05:00

Biology, 24.08.2019 05:00


Mathematics, 24.08.2019 05:00




English, 24.08.2019 05:00

Social Studies, 24.08.2019 05:00


English, 24.08.2019 05:00



Mathematics, 24.08.2019 05:00

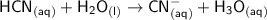
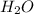 accepts the H⁺ ion ,thus it is a Bronsted-Lowry base.
accepts the H⁺ ion ,thus it is a Bronsted-Lowry base.

