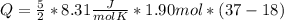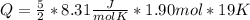Chemistry, 15.08.2020 01:01 coryintheswamp
How much heat (in J) must be added to raise the temperature of 1.90 mol of air from 18.0°C to 37.0°C at constant volume? Assume air is completely diatomic.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1

Chemistry, 23.06.2019 04:20
Calculate the mass of 0.750 mol of the following substance. na3po4.
Answers: 1
You know the right answer?
How much heat (in J) must be added to raise the temperature of 1.90 mol of air from 18.0°C to 37.0°C...
Questions





History, 21.02.2020 19:57





History, 21.02.2020 19:57


Mathematics, 21.02.2020 19:57

Mathematics, 21.02.2020 19:57

Chemistry, 21.02.2020 19:57



Biology, 21.02.2020 19:57

Physics, 21.02.2020 19:57

History, 21.02.2020 19:57

Mathematics, 21.02.2020 19:57

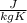 or
or 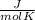 . Two specific heats are defined for gases, one for constant volume (cv) and the other for constant pressure (cp).
. Two specific heats are defined for gases, one for constant volume (cv) and the other for constant pressure (cp).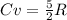 where R is the general gas constant, whose value in this case will be
where R is the general gas constant, whose value in this case will be 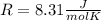 n=1.90 molTfinal= 37 °CTinitial= 18 °C
n=1.90 molTfinal= 37 °CTinitial= 18 °C