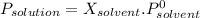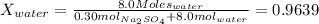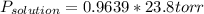Chemistry, 15.08.2020 01:01 nathanwhite2000
at 25*C, the vapor pressure of pure water is 23.8 torr. a solution is prepared by dissolving 0.30 mol Na2SO4 in 8.0 moles of water. what is the vapor pressure of this solution at 25*C

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

Chemistry, 23.06.2019 04:31
2ki + pb(no3)2 → 2kno3 + pbi2 determine how many moles of kno3 are created if 0.03 moles of ki are completely consumed.
Answers: 1

Chemistry, 23.06.2019 09:00
How many grams of ammonia are produced when 1.0 mole of nitrogen reacts
Answers: 2

Chemistry, 23.06.2019 14:00
How is the electron sea model of metallic bonding different from the band theory? how are they the same? give at least one similarity and one difference between the models
Answers: 2
You know the right answer?
at 25*C, the vapor pressure of pure water is 23.8 torr. a solution is prepared by dissolving 0.30 mo...
Questions

History, 03.06.2021 20:40





Mathematics, 03.06.2021 20:40

Mathematics, 03.06.2021 20:40



Mathematics, 03.06.2021 20:40




Geography, 03.06.2021 20:40

Mathematics, 03.06.2021 20:40

Spanish, 03.06.2021 20:40

Mathematics, 03.06.2021 20:40


Mathematics, 03.06.2021 20:40

Social Studies, 03.06.2021 20:40