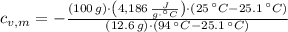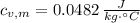Chemistry, 14.08.2020 01:01 TianaTheSquid
A 12.6 g block of a metal intially at a temperature of 94.0 °C is added to a 100.0 g sample of water intially at 21.0 °C. If the final temperature of the water is 25.1 °C, determine the specific heat of the metal. Write all equations or formulas you use, and show as much of your work as you can.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
What is the value of keq for the reaction expressed in scientific notation
Answers: 1

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no]2[o2]
Answers: 3


You know the right answer?
A 12.6 g block of a metal intially at a temperature of 94.0 °C is added to a 100.0 g sample of water...
Questions

History, 30.08.2019 23:30

English, 30.08.2019 23:30


Arts, 30.08.2019 23:30





Mathematics, 30.08.2019 23:30

Social Studies, 30.08.2019 23:30

History, 30.08.2019 23:30


Biology, 30.08.2019 23:30







Business, 30.08.2019 23:30


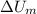 and
and 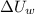 are the changes in internal energies of the block of metal and water, measured in joules.
are the changes in internal energies of the block of metal and water, measured in joules. 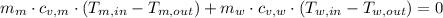
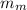 ,
, 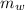 - Masses of the block of metal and water, measured in grams.
- Masses of the block of metal and water, measured in grams.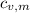 ,
, 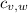 - Specific heats of the block of metal and water, measured in joules per gram-Celsius.
- Specific heats of the block of metal and water, measured in joules per gram-Celsius.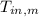 ,
, 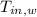 - Initial temperatures of the block of metal and water, measured in Celsius.
- Initial temperatures of the block of metal and water, measured in Celsius. 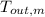 ,
, 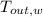 - Final temperatures of the block of metal and water, measured in Celsius.
- Final temperatures of the block of metal and water, measured in Celsius. 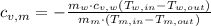
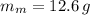 ,
, 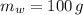 ,
, 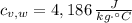 ,
, 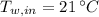 ,
, 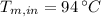 and
and 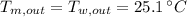 , the specific heat of the metal is:
, the specific heat of the metal is: