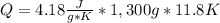Chemistry, 12.08.2020 08:01 runninglovexoxo
Calculate the energy required to heat 1.30kg of water from 22.4°C to 34.2°C . Assume the specific heat capacity of water under these conditions is 4.18·J·g−1K−1 . Round your answer to 3 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:20
Calculate the enthalpy of the following reaction: 4 b (s) + 3 o2 (g) → 2 b2o3 (s) given the following pertinent information: (a) b2o3 (s) + 3 h2o (g) → 3 o2 (g) + b2h6 (g), δhoa = +2035 kj (b) 2 b (s) + 3 h2 (g) → b2h6 (g), δhob = +36 kj (c) h2 (g) + latex: \frac{1}{2} 1 2 o2 (g) → h2o (l), δhoc = −285 kj (d) h2o (l) → h2o (g), δhod = +44 kj
Answers: 3

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2
You know the right answer?
Calculate the energy required to heat 1.30kg of water from 22.4°C to 34.2°C . Assume the specific he...
Questions



Mathematics, 20.01.2021 01:00

Advanced Placement (AP), 20.01.2021 01:00

Mathematics, 20.01.2021 01:00

Mathematics, 20.01.2021 01:00

Social Studies, 20.01.2021 01:00

Social Studies, 20.01.2021 01:00

History, 20.01.2021 01:00

Engineering, 20.01.2021 01:00

Mathematics, 20.01.2021 01:00



Biology, 20.01.2021 01:00


Biology, 20.01.2021 01:00


Arts, 20.01.2021 01:00


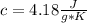 m= 1.30 kg= 1,300 g (1 kg=1,000 g)ΔT= 34.2 °C - 22.4 °C= 11.8 °C= 11.8 °K Being a temperature difference, it is independent if they are degrees Celsius or degrees Kelvin. That is, the temperature difference is the same in degrees Celsius or degrees Kelvin.
m= 1.30 kg= 1,300 g (1 kg=1,000 g)ΔT= 34.2 °C - 22.4 °C= 11.8 °C= 11.8 °K Being a temperature difference, it is independent if they are degrees Celsius or degrees Kelvin. That is, the temperature difference is the same in degrees Celsius or degrees Kelvin.