
Chemistry, 12.08.2020 08:01 Homepage10
Aqueous ammonia is added to a mixture of silver chloride and water. Given that Kf for the reaction between Ag+ and NH3 is large, which of the following are true?
A) The free ions are favored over the complex ion.
B) The complex ion is favored over solid silver chloride.
C) The free Ag+ ion is unstable.
D) More silver chloride will precipitate.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

You know the right answer?
Aqueous ammonia is added to a mixture of silver chloride and water. Given that Kf for the reaction b...
Questions


Biology, 26.05.2020 02:00

History, 26.05.2020 02:00

English, 26.05.2020 02:00



Mathematics, 26.05.2020 02:00

Mathematics, 26.05.2020 02:00




Mathematics, 26.05.2020 02:00

Mathematics, 26.05.2020 02:00

Biology, 26.05.2020 02:00



Mathematics, 26.05.2020 02:00



English, 26.05.2020 02:00

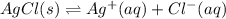
![Ag^+(aq)+2NH_3(aq)\rightleftharpoons [Ag(NH_3)_2]^+(aq)](/tpl/images/0720/0283/481b4.png)


