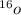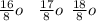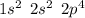. A certain diatomic element is a colorless gas and is highly reactive. It has three isotopes with mass #’s 16, 17 and 18 i. What is the naturally occurring diatomic element ? (2 points) ii. Write the isotopic symbols. (2 points) iii. How many neutrons does each possess? (2 points) iv. What is the electronic configuration of each isotope?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1

Chemistry, 23.06.2019 03:00
What do electromagnetic waves carry? how are they produced through which media can they move? where do they transfer energy? what do they not transfer? what do mechanical waves carry? how are they produced? through which media can they move? where do they transfer energy? what do they not transfer?
Answers: 1
You know the right answer?
. A certain diatomic element is a colorless gas and is highly reactive. It has three isotopes with m...
Questions

Mathematics, 19.09.2019 11:50

Mathematics, 19.09.2019 11:50

Mathematics, 19.09.2019 11:50

Biology, 19.09.2019 11:50


Social Studies, 19.09.2019 11:50


Mathematics, 19.09.2019 11:50




Chemistry, 19.09.2019 11:50

Mathematics, 19.09.2019 11:50

Mathematics, 19.09.2019 11:50


Business, 19.09.2019 11:50

History, 19.09.2019 11:50