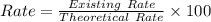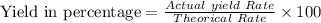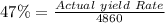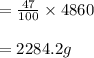Chemistry, 12.08.2020 07:01 hannahmckain
A pharmaceutical company is making a large volume of nitrous oxide (NO). They predict they will be able to make a maximum amount of 4860 grams with the materials they have in stock. From the previous 10 volumes they have made, they know that the percent yield of this reaction is fairly low at 47%. How much will the actual yield be? A. 228 grams B. 2284 grams C. 10340 grams D. 486 grams

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

Chemistry, 22.06.2019 03:50
Express the following number in scientific notation. 0.026890 =
Answers: 1


You know the right answer?
A pharmaceutical company is making a large volume of nitrous oxide (NO). They predict they will be a...
Questions


English, 23.07.2019 05:30


Mathematics, 23.07.2019 05:30








Health, 23.07.2019 05:30



Mathematics, 23.07.2019 05:30

Mathematics, 23.07.2019 05:30

Chemistry, 23.07.2019 05:30


Chemistry, 23.07.2019 05:30