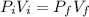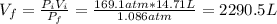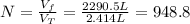A cylinder containing 14.71 L of helium gas at a pressure of 169.1 atm is to be used to fill toy balloons to a pressure of 1.086 atm. Each inflated balloon has a volume of 2.414 L. What is the maximum number of balloons that can be inflated? Report your answer to 1 decimal place. (Remember that 14.71 L of helium at 1.086 atm will remain in the exhausted (empty) cylinder)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Zinc + lead(ii) nitrate yield zinc nitrate + leadwhat's the chemical equation for this?
Answers: 1

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1
You know the right answer?
A cylinder containing 14.71 L of helium gas at a pressure of 169.1 atm is to be used to fill toy bal...
Questions


Mathematics, 14.10.2019 11:30

History, 14.10.2019 11:30

Mathematics, 14.10.2019 11:30

Social Studies, 14.10.2019 11:30

History, 14.10.2019 11:30


Mathematics, 14.10.2019 11:30

History, 14.10.2019 11:30

Biology, 14.10.2019 11:30

Mathematics, 14.10.2019 11:30



Mathematics, 14.10.2019 11:30

Mathematics, 14.10.2019 11:30



Biology, 14.10.2019 11:30

Mathematics, 14.10.2019 11:30

Geography, 14.10.2019 11:30

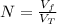
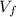 : is the volume at 1.086 atm
: is the volume at 1.086 atm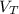 : is the balloon volume = 2.414 L
: is the balloon volume = 2.414 L