
Chemistry, 04.08.2020 14:01 rajeeblagrove
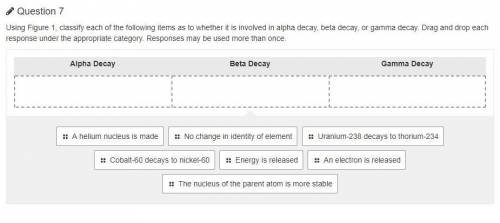
Using Figure 1, classify each of the following items as to whether it is involved in alpha decay, beta decay, or gamma decay. Drag and drop each response under the appropriate category. Responses may be used more than once.


Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 00:00
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1


Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1
You know the right answer?
Using Figure 1, classify each of the following items as to whether it is involved in alpha decay, be...
Questions

Mathematics, 23.10.2020 22:10


History, 23.10.2020 22:10

History, 23.10.2020 22:10

Mathematics, 23.10.2020 22:10



Mathematics, 23.10.2020 22:10




Mathematics, 23.10.2020 22:10








Mathematics, 23.10.2020 22:10



