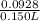Chemistry, 01.08.2020 23:01 alystidham1439
Suppose of nickel(II) iodide is dissolved in of a aqueous solution of potassium carbonate. Calculate the final molarity of nickel(II) cation in the solution. You can assume the volume of the solution doesn't change when the nickel(II) iodide is dissolved in it. Round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
Suppose of nickel(II) iodide is dissolved in of a aqueous solution of potassium carbonate. Calculate...
Questions

English, 22.09.2020 04:01

Mathematics, 22.09.2020 04:01


Health, 22.09.2020 04:01


Mathematics, 22.09.2020 04:01

Mathematics, 22.09.2020 04:01

Mathematics, 22.09.2020 04:01


Mathematics, 22.09.2020 04:01

English, 22.09.2020 04:01

Mathematics, 22.09.2020 04:01

Mathematics, 22.09.2020 04:01


Mathematics, 22.09.2020 04:01




Mathematics, 22.09.2020 04:01

 - 312.5 g
- 312.5 g