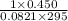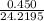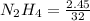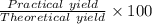Chemistry, 31.07.2020 20:01 gapaxton22
Hydrazine, N2H4 , reacts with oxygen to form nitrogen gas and water. N2H4(aq)+O2(g)⟶N2(g)+2H2O(l) If 2.45 g of N2H4 reacts with excess oxygen and produces 0.450 L of N2 , at 295 K and 1.00 atm, what is the percent yield of the reaction?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 23.06.2019 03:40
Write the overall equation for the reaction occurring in lithium battery?
Answers: 3

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1
You know the right answer?
Hydrazine, N2H4 , reacts with oxygen to form nitrogen gas and water. N2H4(aq)+O2(g)⟶N2(g)+2H2O(l) If...
Questions

Mathematics, 08.07.2019 20:30





History, 08.07.2019 20:30



Biology, 08.07.2019 20:30

Mathematics, 08.07.2019 20:30

Social Studies, 08.07.2019 20:30


Mathematics, 08.07.2019 20:30



History, 08.07.2019 20:30

Spanish, 08.07.2019 20:30

Mathematics, 08.07.2019 20:30


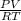
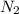 =
=