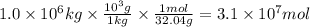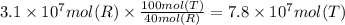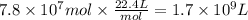Chemistry, 31.07.2020 08:01 daijahamaker062816
Methanol is produced industrially by catalytic hydrogenation of carbon monoxide according to the following equation: CO(g) + 2 H2(g) → CH3OH(l) If the yield of the reaction is 40%, what volume of CO (measured at STP) would be needed to produce 1.0 × 106 kg CH3OH?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Asolution has a ca2+ concentration of 0.049 m and an f- concentration is 0.147 m at equilibrium. the value of ksp for caf2 at 25°c is 4.0 x 10-11. will this solution form a precipitate? yes no
Answers: 3

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1

Chemistry, 21.06.2019 23:30
Why do you suppose the structural polysaccharide cellulose does not contain branches? why do you suppose the structural polysaccharide cellulose does not contain branches? branches in the molecule would generate side chains that would almost certainly make it difficult to pack the cellulose molecules into globules, thereby decreasing the flexibility and strength of the globules. branches in the molecule would generate side chains that would almost certainly make it difficult to pack the cellulose molecules into microfibrils, thereby increasing the rigidity and strength of the microfibrils. branches in the molecule would generate side chains that would almost certainly make it difficult to pack the cellulose molecules into globules, thereby increasing the flexibility and strength of the globules. branches in the molecule would generate side chains that would almost certainly make it difficult to pack the cellulose molecules into microfibrils, thereby decreasing the rigidity and strength of the microfibrils.
Answers: 1

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1
You know the right answer?
Methanol is produced industrially by catalytic hydrogenation of carbon monoxide according to the fol...
Questions

Mathematics, 18.10.2021 21:20


Advanced Placement (AP), 18.10.2021 21:20

Mathematics, 18.10.2021 21:20

History, 18.10.2021 21:20


Mathematics, 18.10.2021 21:20

Mathematics, 18.10.2021 21:20


Mathematics, 18.10.2021 21:20

Arts, 18.10.2021 21:20


Mathematics, 18.10.2021 21:20

Mathematics, 18.10.2021 21:20

Chemistry, 18.10.2021 21:20


SAT, 18.10.2021 21:20


Mathematics, 18.10.2021 21:20