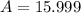In nature, oxygen has three common isotopes. The atomic masses and relative abundances of these isotopes are given in the table below. Isotope Atomic Mass (amu) Relative Abundance O-16 15.995 99.759% O-17 16.995 0.037% O-18 17.999 0.204% Calculate the average atomic mass of oxygen. Show all of your calculations below.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:10
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
In nature, oxygen has three common isotopes. The atomic masses and relative abundances of these isot...
Questions


History, 19.05.2021 07:20

Mathematics, 19.05.2021 07:20



Mathematics, 19.05.2021 07:20

Health, 19.05.2021 07:20

Chemistry, 19.05.2021 07:20

Mathematics, 19.05.2021 07:20

Mathematics, 19.05.2021 07:20






Computers and Technology, 19.05.2021 07:20

Mathematics, 19.05.2021 07:20


English, 19.05.2021 07:20

Mathematics, 19.05.2021 07:20

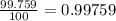
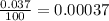
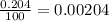
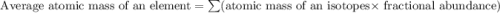
![A=\sum[(15.995\times 0.99759)+(16.995\times 0.00037)+(17.999 \times 0.00204)]](/tpl/images/0715/9581/687c5.png)