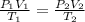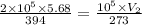Chemistry, 30.07.2020 05:01 andresduenas72
A pressure cooker contains 5.68 liters of air at a temperature of 394 K. If the absolute pressure of the air in the pressure cooker is 205 pascals, how many moles of air are in the cooker? The cooker contains moles of air. 1 SEE ANSWER

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 23.06.2019 00:00
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1
You know the right answer?
A pressure cooker contains 5.68 liters of air at a temperature of 394 K. If the absolute pressure of...
Questions

Mathematics, 24.02.2021 18:40

History, 24.02.2021 18:40




Mathematics, 24.02.2021 18:40


English, 24.02.2021 18:40

Mathematics, 24.02.2021 18:40


Mathematics, 24.02.2021 18:40




Mathematics, 24.02.2021 18:40

Social Studies, 24.02.2021 18:40


Chemistry, 24.02.2021 18:40