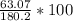Chemistry, 30.07.2020 03:01 christinafish9303
Aspirin (C9H8O4) is produced by the reaction of salicylic acid (C7H6O3, Molar mass = 138.1 g/mol) and acetic anhydride (C4H6O3, Molar mass = 102.1 g/mol) based on the BALANCED equation : C7H6O3(s) + C4H6O3(l ) → C9H8O4(s) + C2H4O2( l) If 63.07 grams of aspirin (Molar mass = 180.2 g/mol) was collected from an experiment when 138.1 grams C7H6O3 reacted with excess C4H6O3, what was the percent yield?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

You know the right answer?
Aspirin (C9H8O4) is produced by the reaction of salicylic acid (C7H6O3, Molar mass = 138.1 g/mol) an...
Questions

Mathematics, 05.03.2021 01:10



Mathematics, 05.03.2021 01:10

Biology, 05.03.2021 01:10





Mathematics, 05.03.2021 01:10


Mathematics, 05.03.2021 01:10

Mathematics, 05.03.2021 01:10


Mathematics, 05.03.2021 01:10


Chemistry, 05.03.2021 01:10

Mathematics, 05.03.2021 01:10

Mathematics, 05.03.2021 01:10

Mathematics, 05.03.2021 01:10

 hould be used up in the reaction, yielding 180.2 g of C9H8O4. ⇒ Theoretical yield = 180.2g
hould be used up in the reaction, yielding 180.2 g of C9H8O4. ⇒ Theoretical yield = 180.2g