
Write a balanced equation for the single-replacement oxidation-reduction reaction described, using the smallest possible integer coefficients. The reaction that takes place when chlorine gas combines with aqueous potassium bromide. (Use the lowest possible coefficients. Omit states of matter.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2


Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3
You know the right answer?
Write a balanced equation for the single-replacement oxidation-reduction reaction described, using t...
Questions

Biology, 16.09.2019 18:00

Business, 16.09.2019 18:00




Physics, 16.09.2019 18:00


Chemistry, 16.09.2019 18:00




History, 16.09.2019 18:00

Mathematics, 16.09.2019 18:00


Mathematics, 16.09.2019 18:00

History, 16.09.2019 18:00

Social Studies, 16.09.2019 18:00



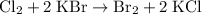 .
.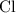 and bromine
and bromine 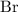 are group 17 elements (halogens.) Each On the other hand, potassium
are group 17 elements (halogens.) Each On the other hand, potassium 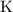 is a group 1 element (alkaline metal.) Each
is a group 1 element (alkaline metal.) Each  . Similarly, the ratio between
. Similarly, the ratio between 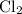 , while the formula for bromine gas is
, while the formula for bromine gas is 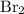 .
.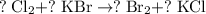 .
.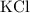 features two elements each.
features two elements each.  .
.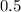 units of
units of 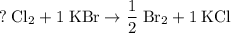 .
.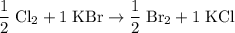 .
.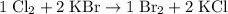 .
.

