
Chemistry, 29.07.2020 16:01 hurtadocrv
Which of the following solutions would have the highest pH? Assume that they are all 0.10 M in acid at 25°C. The acid is followed by its Ka value.
a. HCHO2, 1.8 x 10-4
b. HF, 3.5 x 10-4
c. HClO2, 1.1 x 10-2
d. HCN, 4.9 x 10-10
e. HNO2, 4.6 x 10-4

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Which of the following solutions would have the highest pH? Assume that they are all 0.10 M in acid...
Questions

Mathematics, 19.05.2021 04:50


Mathematics, 19.05.2021 04:50

Mathematics, 19.05.2021 04:50


English, 19.05.2021 04:50

Biology, 19.05.2021 04:50

English, 19.05.2021 04:50


Mathematics, 19.05.2021 04:50



Mathematics, 19.05.2021 04:50


Mathematics, 19.05.2021 04:50




History, 19.05.2021 04:50

Mathematics, 19.05.2021 04:50

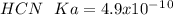
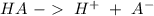
![Ka=\frac{[H^+][A^-]}{[HA]}](/tpl/images/0714/8162/39962.png)
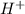 . Therefore, if we have a higher Ka value we will have a smaller pH (lets keep in mind that with a higher
. Therefore, if we have a higher Ka value we will have a smaller pH (lets keep in mind that with a higher 

