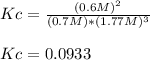Chemistry, 29.07.2020 04:01 sparky1234
The Haber Process is the main industrial procedure to produce ammonia. The reaction combines nitrogen from air with hydrogen mainly from natural gas (methane) and is reversible and exothermic. The enthalpy change for this reaction is - 92 kJ mol-1. In an experiment, 1.5 moles of N2 and 4.0 moles of H2 is mixed in a 1.50 dm3 reaction vessel at 450 °C. After reaching equilibrium, the mixture contained 0.9 mole of NH3.
A) With the above information, write the reaction equilibrium equation in the Haber process. t.
B) Calculate Kc for this reaction.
C) What is the equilibrium yield of ammonia in this reaction?
D) Referring to Le Chatelier's principle and above information, suggest two ways to increase the yield of ammonia in this reaction and explain.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 02:00
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3
You know the right answer?
The Haber Process is the main industrial procedure to produce ammonia. The reaction combines nitroge...
Questions

English, 21.01.2021 19:30

History, 21.01.2021 19:30

Health, 21.01.2021 19:30



English, 21.01.2021 19:30


English, 21.01.2021 19:30

Computers and Technology, 21.01.2021 19:30


German, 21.01.2021 19:30



Mathematics, 21.01.2021 19:30


Mathematics, 21.01.2021 19:30

Social Studies, 21.01.2021 19:30

Mathematics, 21.01.2021 19:30

Physics, 21.01.2021 19:30

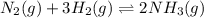 .
.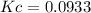 .
.![Kc=\frac{[NH_3]^2}{[N_2][H_2]^3}](/tpl/images/0714/5909/3f3ab.png)
 , we have:
, we have:![[NH_3]=0.6M=2*x](/tpl/images/0714/5909/a9b24.png)
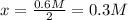
![[N_2]=\frac{1.5mol}{1.5L}-x=1M-0.3M=0.7M](/tpl/images/0714/5909/a34c9.png)
![[H_2]=\frac{4mol}{1.5L}-3*x=2.67M-0.9M=1.77M](/tpl/images/0714/5909/69c53.png)