
In each of the following groups of substances, pick the one that has the given property. Justify your answer. a) highest boiling point: HBr, Kr, Cl2 b) highest freezing point: H2O, NaCl, Hf c) lowest vapor pressure at 25C: Cl2, Br2, I2 d) lowest freezing point: N2, CO, CO2 e) lowest boiling point: CH4, CH3CH3, CH3CH2CH3 f) highest boiling point: HF, HCl, HBr Could someone help me understand fully how to do this?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2

You know the right answer?
In each of the following groups of substances, pick the one that has the given property. Justify you...
Questions

Mathematics, 04.12.2020 17:50



Mathematics, 04.12.2020 17:50





Arts, 04.12.2020 17:50

Health, 04.12.2020 17:50

History, 04.12.2020 17:50


Mathematics, 04.12.2020 17:50

Chemistry, 04.12.2020 17:50






Health, 04.12.2020 17:50

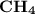 has the lowest molecular weight, thus the lowest freezing point.
has the lowest molecular weight, thus the lowest freezing point.

