
To determine the concentration of chloride ion (Cl-) in a 100 mL sample of ground water, a chemist adds a large enough volume of AgNO3 solution to precipitate all Cl- as AgCl. The mass of the resulting precipitate is 93.9 mg. What is the chloride ion concentration in milligrams of chloride per liter of ground water

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
To determine the concentration of chloride ion (Cl-) in a 100 mL sample of ground water, a chemist a...
Questions


Mathematics, 08.02.2021 17:30






English, 08.02.2021 17:30

Physics, 08.02.2021 17:30


Mathematics, 08.02.2021 17:30

History, 08.02.2021 17:30


Biology, 08.02.2021 17:30


Biology, 08.02.2021 17:30

Biology, 08.02.2021 17:30



Mathematics, 08.02.2021 17:30

![[Cl^-]=232.3\frac{mgCl^-}{L}](/tpl/images/0713/4238/a2cee.png)
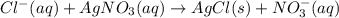
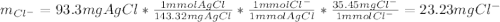
![[Cl^-]=\frac{23.2mgCl^-}{0.100L}\\](/tpl/images/0713/4238/dfd21.png)


