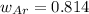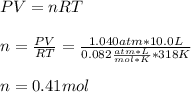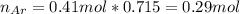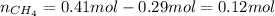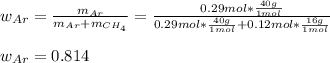Chemistry, 26.07.2020 01:01 jaredsangel08
A 10.0 L flask at 318 K contains a mixture of Ar and CH4 with a total pressure of 1.040 atm. If the mole fraction of Ar is 0.715, what is the mass percent of Ar?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2
You know the right answer?
A 10.0 L flask at 318 K contains a mixture of Ar and CH4 with a total pressure of 1.040 atm. If the...
Questions

Mathematics, 18.03.2021 01:00

Arts, 18.03.2021 01:00


English, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Social Studies, 18.03.2021 01:00

History, 18.03.2021 01:00





Mathematics, 18.03.2021 01:00

Spanish, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00




Mathematics, 18.03.2021 01:00