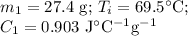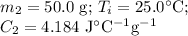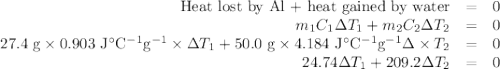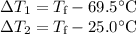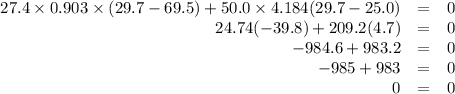Chemistry, 25.07.2020 20:01 kristieroth1
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initially at 25.0 °C and allowed to reach thermal equilibrium. What is the final temperature of the aluminum and water, given that the specific heat of aluminum is 0.903 J/(g·°C)? Assume no heat is lost to surroundings.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1


You know the right answer?
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initi...
Questions


Chemistry, 03.06.2021 17:40


Mathematics, 03.06.2021 17:40

Social Studies, 03.06.2021 17:40



English, 03.06.2021 17:40


Mathematics, 03.06.2021 17:40


Biology, 03.06.2021 17:40

History, 03.06.2021 17:40

Mathematics, 03.06.2021 17:40


Spanish, 03.06.2021 17:40



Computers and Technology, 03.06.2021 17:40